In 2018, I was asked to be a patient advocate for Biogen for their Alzheimer drug aducanumab, unfortunately they cancelled trials. Biogen’s stocks also fell last March when the trials for aducanumab- (the only drug that removes plaque from the brain in Alzheimer’s patients) had displayed little efficacy.

I was shocked, as I followed the trials that were taking place and some patients were doing well. This alone was hopeful. The only drug available for Alzheimer’s patients is Aricept for mild to moderate Alzheimer’s disease and Memantine -for moderate to severe Alzheimer’s disease- and neither drug removes plaque (which is the sticky protein that attaches to the brain and kills neurons). Both drugs only slow down the progression to make caregiving a bit easier- however both drugs have severe side effects- and if I knew that prior to my mom’s disease- I would not have given her either drug. (She was off of medication for 10 years- once I found out about the side effects) She was only on Vitamins.

Biogen partnered with Esai Co to develop aducanumab and when they announced in October 2019 that they wanted to revive the drug, their stocks rose almost to 11 billion. Some patients have benefited from the aducanumab. This to me is hopeful. “It all comes down to FDA’s willingness to approve a novel therapy with a debatable dataset in a disease that affects millions of patients,” said Guggenheim Securities analyst Yatin Suneja.
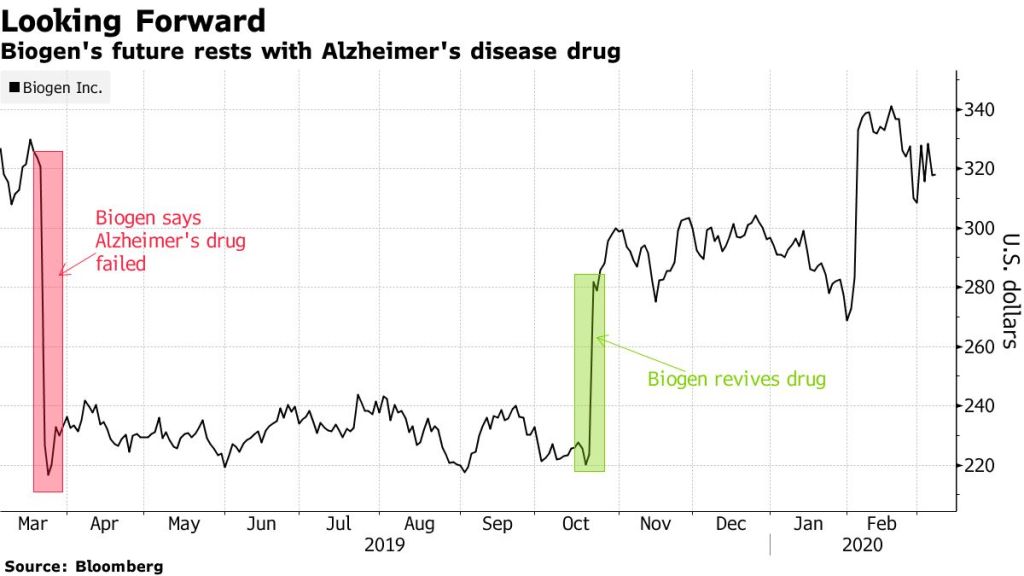
It is now left up to the FDA on whether they will approve this drug. Biogen was also working on BAN2401 another Alzheimer drug – when I spoke to my contact at Biogen- 2 years ago, they were dedicated to finding a treatment for the disease and were not giving up.

Biogen has been the only company that is aggressively endeavoring to develop drugs for diseases that attack the brain and central nervous system. Biogen also signed a $75 million deal to buy Pfizer Inc. another drug candidate for Alzheimer’s in January.
Investors and analysts are saying that the verdict may come down to politics. This would be a big win in an election year, if the FDA approves aducanumb.

A decision could come around the November election, if Biogen is granted a priority review. Biogen is among the five largest U.S Based biotechnology companies with $55 billion in market value. BlackRock, PrimeCap Management and Vanguard Group have big positions in the company and are heavyweight investment advisors. AQR Capital Management, Arrowstreet Capital and DE Shaw & Co. hedge funds have millions riding on Biogen’s success.

If aducanumab gets FDA approval, it could reignite interest in other companies that could not show efficacy with other pharmaceuticals that targeted the amyloid plaque in the brain. I for one, am rooting for this drug, it’s been a long time coming – we need a cure!!!!

